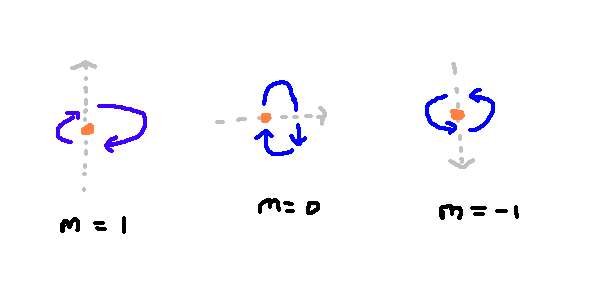Explaining Quantum Degeneracy
How do we even begin to organise the possible states a quantum system can have?
How do we even begin to organise the possible states a quantum system can have?
In quantum mechanics, physical properties like energy only can live in discrete values. If quantities like these are discrete, they are significantly easier to count and understand because the math is much less complicated. For example, studying a system with energy levels labelled by E = 1, 2, 3.. and so on is a whole lot easier than describing a system with a continuum of energy levels with a complex pattern! Therefore, in physics, it is feasible and essential to try and classify the different discrete states that a physical system can live in. This attempt to organise things raises many interesting questions.
A quantum ‘eigenstate' is a certain configuration of a physical system that we can actually observe through an experiment. It is a physically possible 'state of being' that we can watch, for example, in a hydrogen atom (or any other physical system) once we measure the atom. The simplest example is that of a coin. In quantum mechanics, we know that objects behave probabilistically. If I flip a coin, there are two possible states that it could land in, but a priori, we don't know which one it's going to be in until we do the experiment.
The example of a coin is pretty much the simplest expression of what a quantum state is. We have a physical system that is governed by a random distribution, but when we observe it, it spits out a quantum state that we can observe.
There are many different states that we can observe a physical system in — the question is then, how do we organise all of these physical states in a way that makes sense?
Are there any other physical quantities that necessarily live in discrete values (spoiler: yes)?
Suppose we have two physical observables A and B, and that both of these observables can only take quantities in discrete values. Is there a physical state for every single combination of values that A and B can collectively take? What is the structure of these physical quantities?
How many physical observables do we need to label all the different sets of quantum states completely?
A weird, unexplained anomaly in Hydrogen's energy levels
In a previous post, I explained the role of quantum mechanics in modelling the different energy levels of Hydrogen. Using Schrodinger's equation as a model, we concluded that the electron in a Hydrogen atom couldn't just live in a continuum of different states. Instead, it can only live in a discrete, ladder-like selection of different energies.
I also mentioned that energy was not the only thing that lived in discrete values. There are other important physical quantities, which you may have heard from popular science books, that only live in discrete levels. One of these quantities is called angular momentum. What exactly is angular momentum? You may know from high school that momentum measures how hard an object is to stop if it is moving. This type of momentum has a name, but it is typically omitted — linear momentum. Instead, it is calculated as the mass times the velocity of the object.
Angular momentum is precisely the same but applies to rotations. It measures how difficult it is to stop an object from rotating around a given origin.
The diagram above shows the difference between linear and angular momentum. For linear momentum, the particle is modelled to be moving in a straight line. However, for angular momentum, the picture is slightly tricker. As you can see in the diagram, a single particle is rotating around an axis in the centre of the circle. The angular momentum is denoted by the letter l, and it points towards the axis of rotation. Mathematically, we say it is the cross product of the vector pointing to it from the origin and the vector of its linear momentum at that point in its trajectory.
It turns out that electrons can occupy different possible angular momentum states within a single discrete energy level. Basic quantum mechanics also correctly predicts that electrons can only live within discrete values of angular momentum — precisely this measure of momentum due to a particle's rotation.
The possible states of Hydrogen
From here on out, to avoid confusion, I'll call this model the Bohr Model. In the Bohr model, we have only one possible state that an electron can live in at the lowest energy level. In the second level, we actually see two possible states that an electron can live in. Each of these two states represents a particular angular momentum value that an electron can have. The higher the energy level, the more angular momentum states there are. This is because states with more angular momentum require more energy to keep the atoms spinning.
In the diagram below, you can see that we only have a single state in the first level of energy. In the second level, we are allowed four states. In higher energy levels, we are allowed even more different quantum states. At each given energy level, the other quantum states are labelled by the electron's angular momentum. For historical reasons, we use the letter l as the symbol for this label. In quantum mechanics, the concept of multiple states associated with a single energy level is called degeneracy. Degeneracy occurs when we have numerous different states occupying a given energy level².
Let's explain the colour coding scheme here. We have the first three possible energy levels of Hydrogen, shown in the grey dotted lines. In the first level, calculations with the Bohr equation show that at the lowest energy, we only have a single state with an angular momentum l=0. The state is represented by the dot coloured in red.
When we move to the second energy level, things get slightly more complicated. We have one state which can have 0 total angular momentum — it doesn't rotate. But, another angular momentum level is labelled l=1, which denotes a slightly larger but non-zero angular momentum. In the diagram, this state is shown in yellow. To make things somewhat more complex, within this single angular momentum level of l=1, we also have three states with the same total angular momentum. Each of these states represents the three different possible axes of rotation of the atom.
In the case of the first angular momentum level, I've drawn out each of the three states here.
So, in the Bohr model, It turns out that these properties are all we need to fully describe the quantum state of an electron in a hydrogen atom. In total, we have three numbers that completely define the state of an electron floating around. The first number is energy. The second number is the total angular momentum of this electron atom. The third number is how tilted the rotation is. These numbers are called quantum numbers, and mathematicians typically denote them with a funny looking expression.
How do we push the model further?
The model of Hydrogen by no means stops there. To incorporate Einstein's theory of special relativity, Dirac tried to develop a relativistic Schrodinger equation as an improvement to the Bohr model of Hydrogen. With this, Dirac realised that there was another property called 'spin' that an electron could occupy in addition to the first three properties.
Furthermore, the predicted energy levels are slightly different from what was previously claimed by the Bohr model. But experimentally, they looked correct! However, the story does not stop there, and I will discuss the Dirac equation in my next post.
References
[1] Merzbacher, Eugen (1998). Quantum Mechanics (3rd ed.). New York: John Wiley. ISBN 0471887021.